UK NMD Coordination
The JWMDRC clinical research and network teams have over 15 years experience of setting up and running clinical trials in the neuromuscular field. As coordinators of the EU funded TREAT-NMD Network of Excellence we have been actively involved in re-shaping the research environment for rare inherited neuromuscular diseases, with several members of the team being involved since the beginning.
With specific expertise in running over 30 DMD and SMA clinical trials we are involved in leading and collaborating on national initiatives such as the DMD Hub in order to share the expertise and lessons learnt, coordinate capacity and recruitment at sites and to provide a consistent and professional service to industry.
The experienced multi-disciplinary team within the JWMDRC ideally placed to work with industry, clinical research organisations, and academics to provide advice and guidance and to facilitate the national coordination and set up of trials in the UK for neuromuscular diseases.
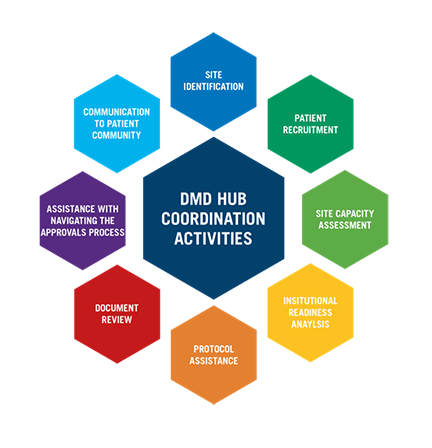
Coordination for neuromuscular clinical trials
- Trial site identification and institutional readiness
- National assessment and coordination of site capacity
- Patient recruitment
- Training and educations events for NMD research teams
- Protocol and IRAS document review
- Assistance with navigating the IRSA/ethics approval processes
- Coordinated communication with patient community and trial sites
Standards of Care

Facilitating improvements and implementation of Standards of Care
The John Walton Muscular Dystrophy Research Centre is working closely with clinical specialists and patient organisations to raise awareness and understanding as well as promote implementation of the latest care recommendations in neuromuscular diseases.
For Duchenne muscular dystrophy, we are doing this as part of an ambitious, 3-year project in collaboration with the UK’s North Star network and with Duchenne UK, Duchenne Research Fund and Joining Jack. We have established specialist working groups looking at the needs, the evidence and the expert opinion across all aspects of the disease. www.dmdcareuk.org
- Support translation of International Standards of Care into accessible Family Guides in multiple languages in collaboration with TREAT-NMD
- Work with specialist centres in the UK to reach consensus on UK-specific standards
- Consult with international colleagues to share experience and best practice
- Identify and address barriers to implementation of expert recommendations
- Work with professional bodies to seek endorsement and buy-in to consensus guidelines
- Identify research questions and seek collaborators and funding to work on them to support Standards of Care
- Support and plan education of the patient and clinical communities about the latest recommendations